Home >> Electricity, E.M.F. , internal resistance
single cell |
The Single Cell
E.M.F.(E) is the p.d. across a cell when it delivers no current.
It can also be thought of as the energy converted into electrical energy, when 1 Coulomb of charge passes through it.
The internal resistance(r) of a cell is a very small resistance. For a 'lead-acid' cell it is of the order of 0.01 Ω and for a 'dry' cell it is about 1 Ω.
This means that a lead-acid cell will deliver a higher current than a dry cell.
We can obtain important equations for E and r by considering a cell with a resistance in a circuit.
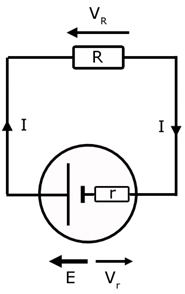
The total resistance Rtotal is the sum of the series resistor and the internal resistance of the cell.
![]()
by summing p.d. around the circuit ,
![]()
substituting for Rtotal
![]()
by Ohm's law, substituting IR = VR
![]()
Note, VR is called the terminal p.d. . That is the p.d. across the cell when it is delivering current.
Measurement of E & r
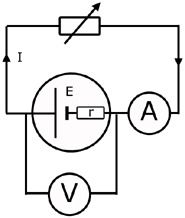
After taking readings of terminal p.d. (VR) and current (I), a graph is drawn.
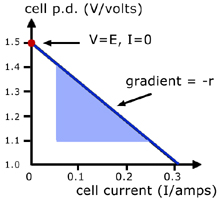
Information can be obtained from the graph by manipulating the equation obtained for E and r:
![]()
![]()
transposing the I and r, turning the equation around,
![]()
comparing with the equation of a straight line,
![]()
Therefore the gradient is '- r' and the intercept on the vertical axis is 'E' .
Cells in Series
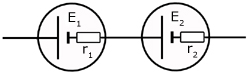
![]()
but
![]()
and
![]()
where r is the internal resistance of the combination
therefore,
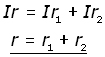
So to sum up, two cells in series are equivalent to one cell with an EMF equal to the sum of the two cells.
The internal resistance of the combination is the sum of the internal resistances of the two cells.
Cells in Parallel
The arrangement dealt with here is only for cells that are similar. For disimilar cells the relationship is complicated, but can be resolved using Kirchhoff's Laws.
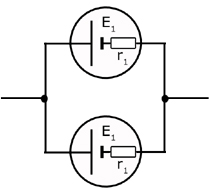
For similar cells the EMF's are equal and the internal resistances are also equal.
Therefore the combined EMF, E is given by,
![]()
and the internal resistance of the combination is calculated from the two internal resistances in parallel:
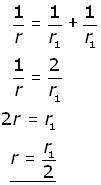
[ About ] [ FAQ ] [ Links ] [ Terms & Conditions ] [ Privacy ] [ Site Map ] [ Contact ]
